Claisen Rearrangement Toward Cyclic Compound on Different Organic Synthesis Methods: Short Review
DOI:
https://doi.org/10.22437/chp.v7i2.26053Keywords:
Cyclic compound synthesis, rearrangement reactions, Claisen Rearrangement, reaction mechanismAbstract
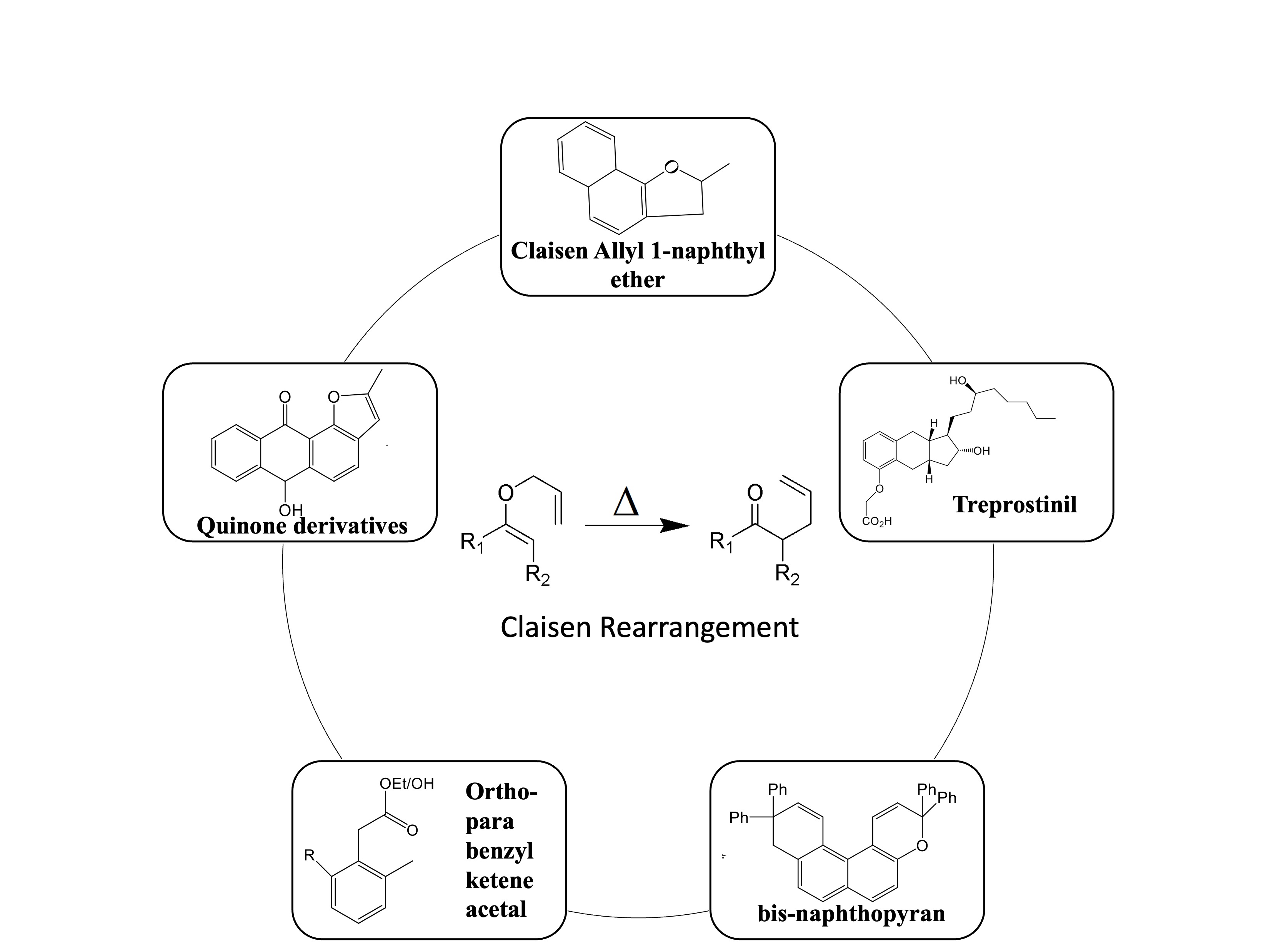
The synthesis of functional and complex organic compounds is majorly performed by the Claisen rearrangement method. Claisen rearrangement is one of [3,3] sigmatropic rearrangements, a complex method in the synthesis of organic compounds, where it is mostly used to construct stereoselective compounds. It can be combined with other synthesis methods to synthesize organic compounds giving satisfactory results based on the method used, temperature, time, and yield produced. This review aimed to summarize several recent advances in synthesizing organic compounds through Claisen rearrangement reactions. An understanding of the mechanism and applications of this reaction might improve the ability to synthesize innovative and useful organic compounds in various fields of life sciences.
Downloads
References
Rao, S., & Prabhu, K. R. (2017). Gold-Catalyzed [2,3]-Sigmatropic Rearrangement: Reaction of Aryl Allyl Alcohols with Diazo Compounds. Organic Letters, 19(4), 846–849. https://doi.org/10.1021/acs.orglett.6b03836
Zhang, L., Hu, M., & Peng, B. (2019). [3,3]- and [5,5]-Sigmatropic Rearrangements of Aryl Sulfoxides Using An ‘Assembly/Deprotonation’ Technology. Synlett, 30(20), 2203–2208. https://doi.org/10.1055/s-0039-1690212
Lin, X., Tan, Z., Yang, W., Yang, W., Liu, X., & Feng, X. (2021). Chiral Cobalt(II) Complex Catalyzed Asymmetric [2,3]-Sigmatropic Rearrangement of Allylic Selenides with α-Diazo Pyrazoleamides. CCS Chemistry, 3(4), 1423–1433. https://doi.org/10.31635/ccschem.020.202000345
Zhu, G., Zhou, J., Liu, L., Li, X., Zhu, X., Lu, X., Zhou, J., & Ye, L. (2022). Catalystâ€Dependent Stereospecific [3,3]â€Sigmatropic Rearrangement of Sulfoxideâ€Ynamides: Divergent Synthesis of Chiral Mediumâ€Sized N , S â€Heterocycles. Angewandte Chemie, 134(28). https://doi.org/10.1002/ange.202204603
Gaykar, R. N., George, M., Guin, A., Bhattacharjee, S., & Biju, A. T. (2021). An Umpolung Oxa-[2,3] Sigmatropic Rearrangement Employing Arynes for the Synthesis of Functionalized Enol Ethers. Organic Letters, 23(9), 3447–3452. https://doi.org/10.1021/acs.orglett.1c00911
Yorimitsu, H., & Perry, G. J. P. (2022). Sulfonium-aided coupling of aromatic rings via sigmatropic rearrangement. Proceedings of the Japan Academy, Series B, 98(4), PJA9804B-03. https://doi.org/10.2183/pjab.98.012
Smith, J. G. (2017). Organic Chemistry (Fifth Edition). MC Graw Hill.
Jukic, M., Sterk, D., & Casar, Z. (2012). Recent Advances in the Retro-Claisen Reaction and Its Synthetic Applications. Current Organic Synthesis, 9(4), 488–512. https://doi.org/10.2174/157017912802651438
Ji, Y.-B., Dong, F., Yu, M., Qin, L., & Liu, D. (2013). Optimization of Synthesis of Seleno-Sargassum fusiforme (Harv.) Setch. Polysaccharide by Response Surface Methodology, Its Characterization, and Antioxidant Activity. Journal of Chemistry, 2013, 1–9. https://doi.org/10.1155/2013/493524
Ripin, D. H. B., & Lewis, C. A. (2020). Rearrangements. In Practical Synthetic Organic Chemistry (pp. 377–423). Wiley. https://doi.org/10.1002/9781119448914.ch7
Ozaki, F., & Okada, Y. (2021). Microwave-assisted Claisen Rearrangement of 1-Allyloxy-4-hydroxybenzene in the Presence of Metal Salt. Current Microwave Chemistry, 8(1), 3–6. https://doi.org/10.2174/2213335607666210106094449
Kotha, S., & Meshram, M. (2018). Application of Claisen Rearrangement and Olefin Metathesis in Organic Synthesis. Chemistry – An Asian Journal, 13(14), 1758–1766. https://doi.org/10.1002/asia.201800613
Tsuda, M., Morita, T., & Nakamura, H. (2022). Synthesis of isoxazoloazaborines via gold(i)-catalyzed propargyl aza-Claisen rearrangement/borylative cyclization cascade. Chemical Communications, 58(12), 1942–1945. https://doi.org/10.1039/D1CC07002A
Song, B. R., Ha, M. W., Kim, D., Park, C., Lee, K. W., & Paek, S.-M. (2019). Investigation of Grignard Reagent as an Advanced Base for Aza-Claisen Rearrangement. Molecules, 24(24), 4597. https://doi.org/10.3390/molecules24244597
Jung, J., Kim, S., & Suh, Y. (2017). Advances in Azaâ€Claisenâ€Rearrangementâ€Induced Ringâ€Expansion Strategies. Asian Journal of Organic Chemistry, 6(9), 1117–1129. https://doi.org/10.1002/ajoc.201700202
Pramesti, I. N., & Okada, Y. (2022). Pengaruh Pelarut Terhadap Reaksi Penataan Ulang Claisen dan Siklisasi Senyawa Alil 1-Naftil Eter dengan Metode Microwave Assisted Organic Synthesis (MAOS). IJCA (Indonesian Journal of Chemical Analysis), 5(2), 86–94. https://doi.org/10.20885/ijca.vol5.iss2.art3
Nadali, S. , Khoshroo, A., & Aghapour, G. (2018). Efficient reductive Claisen rearrangement of prop-2’-enyloxyanthraquinones and 2’-chloroprop-2’-enyloxyanthraquinones with iron powder in ionic liquids. Turkish Journal of Chemistry, 42(3), 883–895. https://doi.org/10.3906/kim-1711-49
Burns, J. M., Krenske, E. H., & McGeary, R. P. (2017). Aromatic Claisen Rearrangements of Benzyl Ketene Acetals: Conversion of Benzylic Alcohols to (ortho-Tolyl)acetates. European Journal of Organic Chemistry, 2017(2), 252–256. https://doi.org/10.1002/ejoc.201601354
Khder, A. E. R. S., Ahmed, S. A., & Altass, H. M. (2016). Mesoporous metal(IV) phosphates as high performance acid catalysts for the synthesis of photochromic bis-naphthopyran via Claisen rearrangement. Reaction Kinetics, Mechanisms and Catalysis, 117(2), 745–759. https://doi.org/10.1007/s11144-015-0963-8
GarcÃa-Lacuna, J., DomÃnguez, G., Blanco-Urgoiti, J., & Pérez-Castells, J. (2019). Synthesis of treprostinil: key Claisen rearrangement and catalytic Pauson–Khand reactions in continuous flow. Organic & Biomolecular Chemistry, 17(43), 9489–9501. https://doi.org/10.1039/C9OB02124H